Question Video: Calculating 𝐾_𝐚 given the Concentration and the Percentage of Dissociation of an Acid | Nagwa
Two springs A and B having spring constant KA and KB (KA = 2KB) are stretched by applying force of equal magnitude. If energy stored in spring A is EA then energy

The dissociation constant (Ka) and the percentage dissociation (alpha) of a weak monobasic acid solution of 0.2 M with a pH = 6 are respectively:
![For the reaction: 2A + B → A2B , the rate = k[A][B]^2 with k = 2.0 × 10^-6 mol^-2 L^2 s^-1 . Calculate the initial rate of the reaction when [A] = For the reaction: 2A + B → A2B , the rate = k[A][B]^2 with k = 2.0 × 10^-6 mol^-2 L^2 s^-1 . Calculate the initial rate of the reaction when [A] =](https://d1hhj0t1vdqi7c.cloudfront.net/v1/TVhJa2FqVWNSMFk=/sd/)






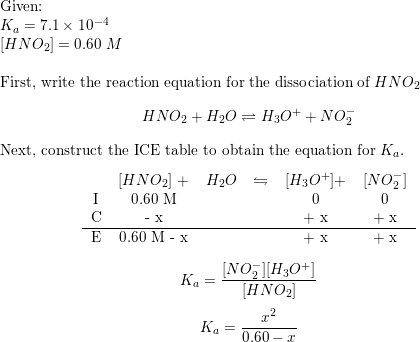






![Calculating [H+] and pH from Ka Calculating [H+] and pH from Ka](https://www.mi.mun.ca/users/pfisher/chemistry1011_134/img013.gif)